Gene Therapy Strategies for Retinitis Pigmentosa
What is Retinitis Pigmentosa?
Retinitis Pigmentosa is a neurodegenerative disease of the retina that leads to blindness due to the loss of the light sensing rod and cone photoreceptors. The disease is caused by genetic mutations in genes expressed in photoreceptors and is therefore an inherited form of retinal degeneration. Interestingly, many patients affected by Retinitis Pigmentosa have mutations in genes that are mainly expressed in rod photoreceptors, which are the night active light sensing cell. However, once most rods have died due to the mutant gene, the death of the day active cones follows. Because cones are essential for color and high acuity vision it is their loss that leads to blindness.
Dr. Claudio Punzo, PhD
A leading researcher for Retinitis Pigmentosa
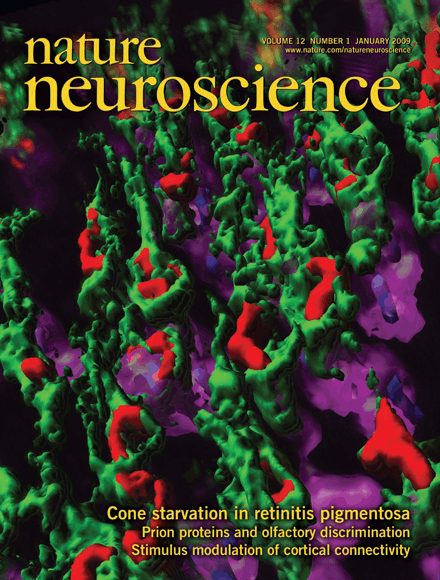
Claudio Punzo, Ph.D., is an Assistant Professor at Department of Ophthalmology, Neurobiology and a faculty member at the Horae Gene Therapy Center at the University of Massachusetts Medical School. Dr. Punzo received his Ph.D. from the University of Basel Switzerland working on Drosophila eye development in the laboratory of Dr. Walter J. Gehring at the Biozentrum. Thereafter he joined the laboratory of Dr. Constance L. Cepko in the Department of Genetics at Harvard Medical School for a post-doctoral fellowship on retinal degeneration. Together with Dr. Cepko he proposed a new paradigm-shifting model for why cones die in Retinitis Pigmentosa even when the mutation is in a gene that is expressed exclusively in rods. The seminal publication, which made the cover story of Nature Neuroscience (see to the left) proposed that cones are slowly starving to death. The goal of the Punzo laboratory is to delay vision loss in Retinitis Pigmentosa by developing a unifying gene therapy strategy to prolong cone survival in the absence of rods.
Dr. Punzo's gene therapy strategy on Retinitis Pigmentosa
Our research has shown that in Retinitis Pigmentosa cones suffer from a nutrient shortage during the periods of cone degeneration, in particular a shortage of glucose. Additionally, we have shown that stimulation of the insulin/mTOR pathway, specifically the kinase mTORC1, results in efficient long-term cone protection to the extent that the approach is therapeutically relevant to humans. Finally, we have shown that the approach we are pursuing is mutation independent meaning that it can help any patient with a mutation in a rod specific gene, in addition to patients where rod death is much faster than cone death even if such patient would have a mutation in a gene that is expressed in both photoreceptor cell types.
The laboratory is currently testing target genes of mTORC1 that can be used for a gene therapeutic approach to prolong vision in Retinitis Pigmentosa since the proof of concept that the approach is valid has been established with genetic mouse models. To this end Dr. Punzo has secured funding from the National Eye Institute, The International Retinal Research Foundation and the Massachusetts Lions Eye Research Fund.
Learn more about the cutting-edge research of Dr. Punzo.
Help us develop a cure!
|
Claudio Punzo, PhDAssociate Professor, Department of Ophthalmology, Neurobiology and Horae Gene Therapy Center, University of Massachusetts Medical School |