Gene Therapy Strategies for Alpha-One Antitrypsin Deficiency
What is alpha-one antitrypsin deficiency?
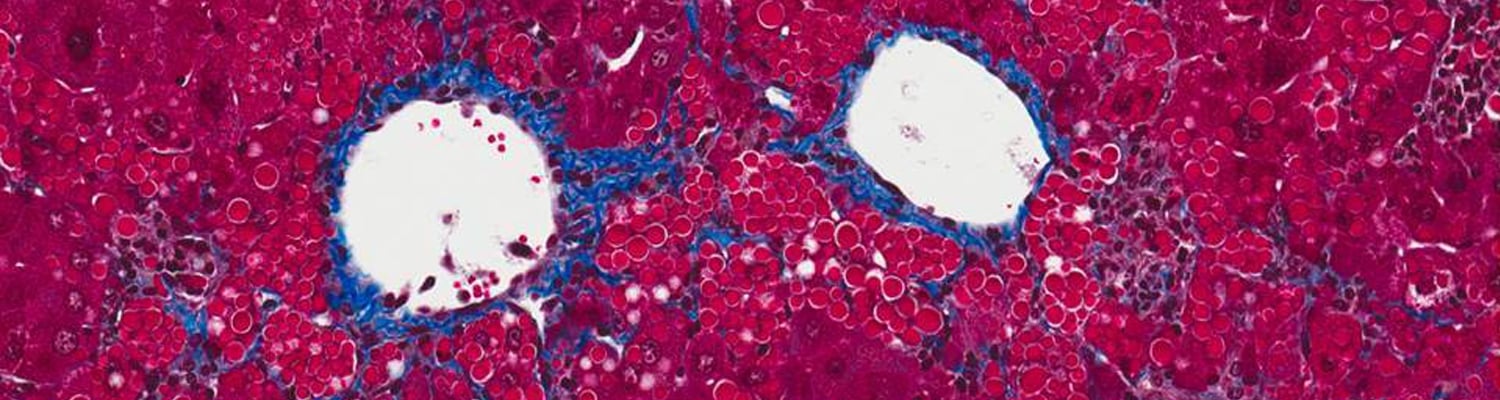
Alpha-one antitrypsin (AAT) deficiency is a common (although often unrecognized) genetic disease, with up to 4% of the population carrying an abnormal AAT gene. Patients can suffer from either a lung disease, which is caused by a loss of the normal AAT action, or from a liver disease, which is caused by build-up of the abnormal mutant protein (called Z-AAT) within the liver cells.
AAT deficiency occurs worldwide, but the prevalence is variable depending on regions. It is a rare disease in Europe and in the US. It affects between 1:1500 and 1:3500 in people of European descent. It is the 2nd most common genetic disorder in Ireland, where 1:25 people carry the mutant gene. It is rare in Asians.
Dr. Terence Flotte, MD
A leading researcher for the alpha-one antitrypsin deficiency
Terence R. Flotte, MD, is the Celia and Isaac Haidak Professor of Medical Education, executive deputy chancellor, provost and dean of the T.H. Chan School of Medicine at UMass Chan Medical School. In 1995, he was the PI of the first human use of rAAV, in a cystic fibrosis (CF) clinical trial. Since then, his lab has conducted several Phase 1 clinical trials for AAT and CF as well as a Phase 2 clinical trial in AAT with vectors developed in his lab. He has received numerous awards, including the 2005 Society for Pediatric Research’s E. Mead Johnson Award for Outstanding Scientific Contributions.
He currently serves as Editor-in-Chief of Human Gene Therapy (and its associated periodicals Human Gene Therapy Methods and Human Gene Therapy Clinical Development) and as the Chair of the Advisory Council of the American Society of Cell and Gene Therapy setting policy and goals for promoting gene therapy research through public education, scientific meetings and scientific committees. Flotte is the author of more than 230 scholarly papers. His research has been funded continuously by the NIH for 22 years.
Dr. Flotte's gene therapy strategy on AAT Deficiency
Flotte's lab currently focuses on systematically developing RNAi-based approaches to Z-AAT down-regulation within hepatocytes and devising appropriate strategies that might allow this to be combined with M-AAT gene augmentation.
Dr. Flotte and his lab members have successfully developed two different investigational clinical gene therapy products for gene augmentation of AAT as a potential therapy for the lung disease (rAAV2-AAT and rAAV1-AAT), and a third vector in development that is much more efficient for delivery of wild-type (M) AAT to hepatocytes (rAAV8-AAT).
|
Terence R. Flotte, MDCelia and Isaac Haidak Professor of Medical Education, Executive Deputy Chancellor, Provost and Dean, T.H. Chan School of Medicine |

