Pioneering cell-surface marker and flow cytometry assays for staging erythroid progenitors and precursors
We pioneered the use of cell-surface markers CD71 and Ter119 to identify, stage and isolate primary mouse erythroblasts. This allows us to identify erythroid precursors as they undergo differentiation in vivo, at specific, discrete developmental stages, and isolate them for further molecular analysis (Koulnis et al., 2011; Liu et al., 2006; Pop et al., 2010; Socolovsky et al., 2001; Zhang et al., 2003).
More recently, we used single-cell RNA-sequencing of mouse hematopoietic tissue to allowed us, for the first time, to define a flow cytometry sorting strategy that purifies much earlier stages of erythroid differentiation directly from hematopoietic tissue, completely isolating the classically-defined burst-forming (BFU-e) and colony-forming progenitors (CFU-e) (Tusi et al., 2018; Swaminathan et al., 2022).
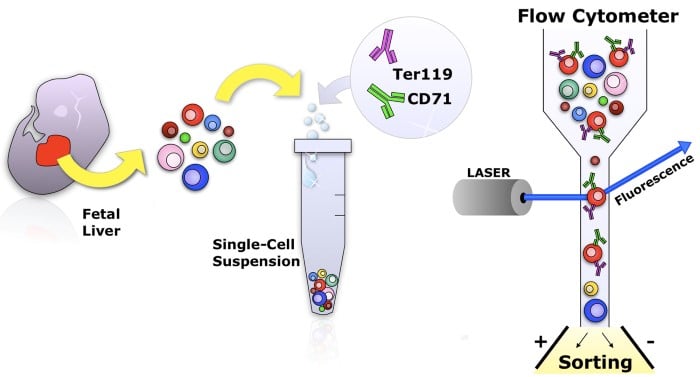
Identification of erythroid progenitors and precursors within mouse fetal liver or other hematopoietic tissue using cell surface markers CD71 and Ter119. The cells can be isolated and analyzed by flow cytometry.
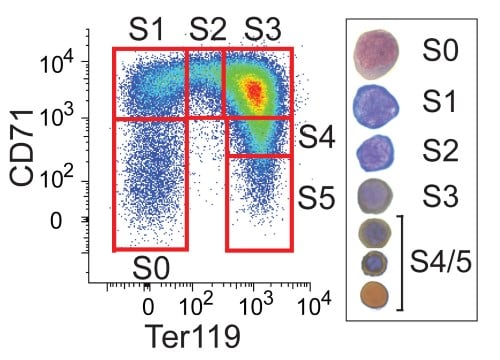
Fetal liver erythroid cells are subdivided into subsets S0 to S5, containing increasingly-mature erythroblasts, based on their CD71/Ter119 expression.
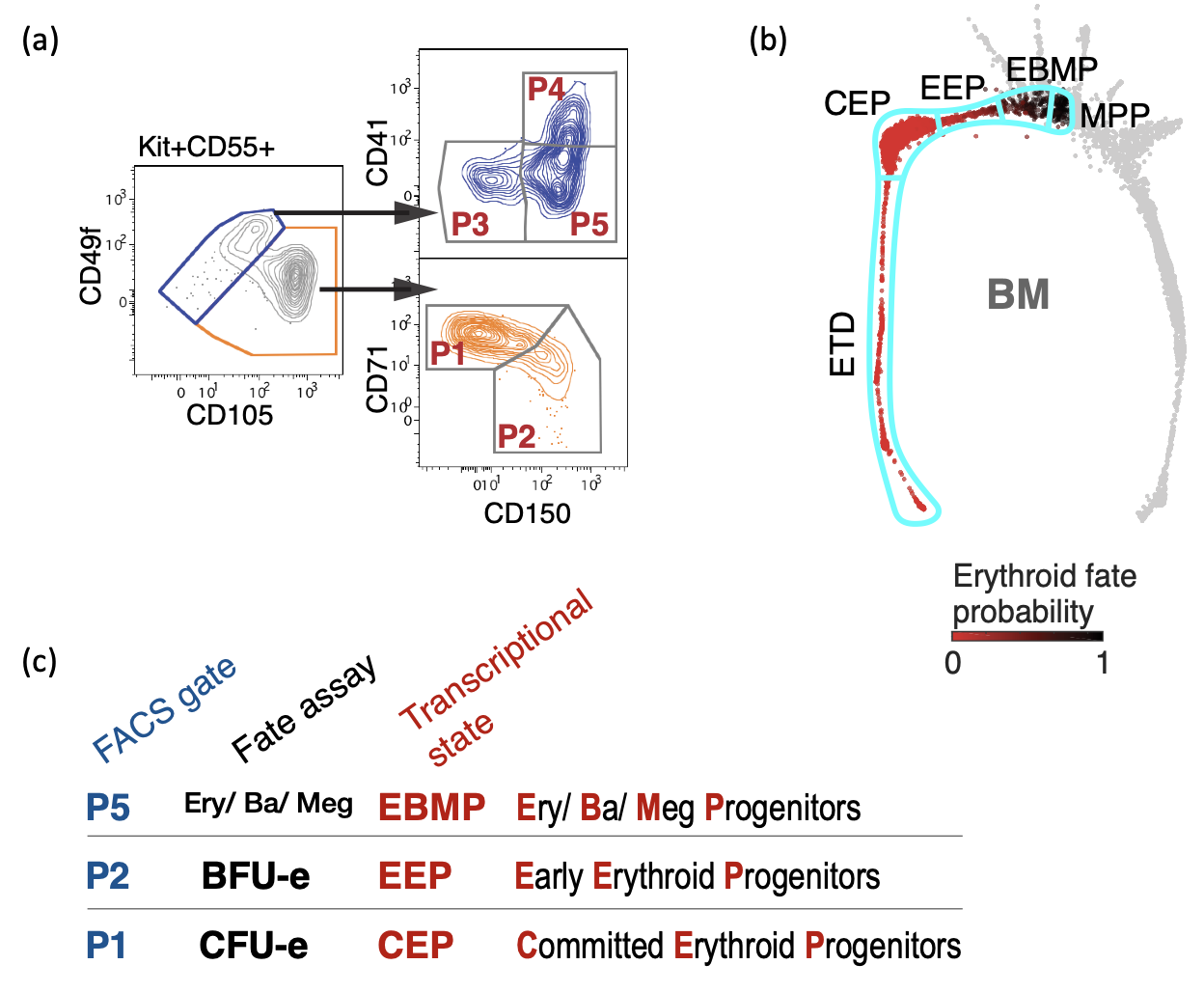
A flow cytometric approach for isolating CFU-e and BFU-e progenitors directly from fresh mouse bone marrow (BM). The flow cytometric populations in (a) correspond to specific transcriptional cell states (b, c).
References:
Koulnis, M., Pop, R., Porpiglia, E., Shearstone, J.R., Hidalgo, D., and Socolovsky, M. (2011). Identification and Analysis of Mouse Erythroid Progenitors using the CD71/TER119 Flow-cytometric Assay. J Vis Exp.
Liu, Y., Pop, R., Sadegh, C., Brugnara, C., Haase, V.H., and Socolovsky, M. (2006). Suppression of Fas-FasL coexpression by erythropoietin mediates erythroblast expansion during the erythropoietic stress response in vivo. Blood 108, 123-133.
Pop, R., Shearstone, J.R., Shen, Q., Liu, Y., Hallstrom, K., Koulnis, M., Gribnau, J., and Socolovsky, M. (2010). A key commitment step in erythropoiesis is synchronized with the cell cycle clock through mutual inhibition between PU.1 and S-phase progression. PLoS Biol 8.
Socolovsky, M., Nam, H., Fleming, M.D., Haase, V.H., Brugnara, C., and Lodish, H.F. (2001). Ineffective erythropoiesis in Stat5a(-/-)5b(-/-) mice due to decreased survival of early erythroblasts. Blood 98, 3261-3273.
Zhang, J., Socolovsky, M., Gross, A.W., and Lodish, H.F. (2003). Role of Ras signaling in erythroid differentiation of mouse fetal liver cells: functional analysis by a flow cytometry-based novel culture system. Blood 102, 3938-3946.
Tusi BK, Wolock SL, Weinreb C, Hwang Y, Hidalgo D, Zilionis R, Waisman A, Huh JR, Klein AM, Socolovsky M. Population snapshots predict early haematopoietic and erythroid hierarchies. Nature. 2018 Mar 1;555(7694):54-60. doi: 10.1038/nature25741. Epub 2018 Feb 21. PMID: 29466336; PMCID: PMC5899604.
Swaminathan A, Hwang Y, Winward A, Socolovsky M. Identification and Isolation of Burst-Forming Unit and Colony-Forming Unit Erythroid Progenitors from Mouse Tissue by Flow Cytometry. J Vis Exp. 2022 Nov 4;(189). doi: 10.3791/64373. PubMed PMID: 36408979.
