UMass Chan Medical School researchers are starting a Phase II clinical trial of Moderna COVID-19 mRNA vaccine boosters in adults. The study comes as scientists chase the continuous emergence of new SARS-CoV-2 virus variants.
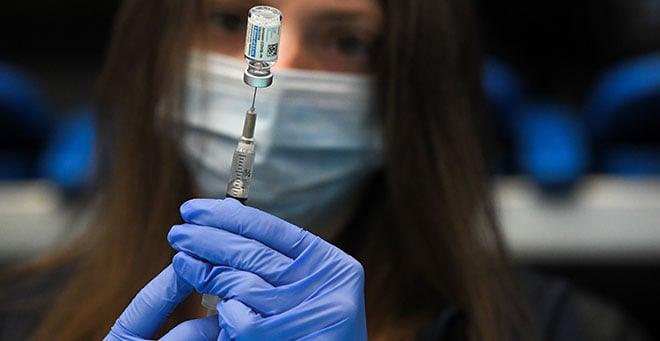
of Moderna’s mRNA vaccine at least six months earlier.
The Moderna study will evaluate different formulations and doses of its boosters among participants. Other studies including one conducted at UMass Chan, which supported the Food and Drug Administration’s emergency use authorization of Pfizer-BioNTech’s COVID-19 vaccine booster, tested the safety and ability to boost immune response of a third standard dose of the vaccine.
Jennifer Wang, MD, professor of medicine, said participants will be randomly assigned to one of six arms: one group will receive the standard Moderna mRNA vaccine booster dose; three groups will receive a new formulation of the mRNA vaccine, which covers major parts of the spike protein, at one of three different dosage levels; and two groups will receive different doses of a combination vaccine that covers both the original spike protein and the beta variant spike protein.
The study is expected to start at UMass Chan on Dec. 20. An anticipated 420 adults will be enrolled across 13 sites nationwide. Participants must be at least 18 years old and have received their second dose of Moderna’s mRNA vaccine at least six months earlier. People who have been recently exposed to someone with COVID-19, or who are pregnant, breastfeeding, or have an unstable chronic disease requiring medical or surgical care are among those who are excluded.
Participants will be followed for one year. Enrollment will be stratified by age, with those 18 to 55 years of age comprising at least 20 percent but not more than 50 percent of the total, and those ages 56 or older.
A key question, according to Dr. Wang, is how well vaccines and boosters will protect against emerging variants. Wang said the omicron variant quickly surged through South Africa, but it wasn’t competing against delta as the dominant strain there. In much of North America and Europe, delta remains predominant and while omicron is spreading, it remains unclear how the epidemiology will evolve.
“We’re learning a little bit more each day and it’s going to take some time,” she said.
Wang’s social media post announcing enrollment in the study, which appeared Dec. 3 on Twitter, has generated a lot of interest.
“I am new to Twitter since COVID, and my feeds are all focused on infectious diseases. It’s the way to get information immediately now, and when it comes from your scientific peers and the greater press, it can be a fantastic resource for keeping up to date with COVID,” she said.
Wang said of her work combatting the spread of disease: “It’s fascinating. It’s exhausting. There’s never a dull moment and there’s something to learn, which is a plus.” And now, influenza is re-emerging in Massachusetts after very low rates last year. “There’s going to be dynamics that will keep us all on our toes,” said Wang.
Those interested in participating in the Moderna booster trial may contact Danielle Kokoski, RN, lead research nurse coordinator II, at danielle.kokoski@umassmed.edu.
Related stories on UMassMed News:
Influenza expected to make a comeback; vaccinations urged
UMass Chan study finds association between long-COVID symptoms and altered oral microbiome
COVID vaccine trial for kids on track, says KidCOVE principal investigator at UMMS