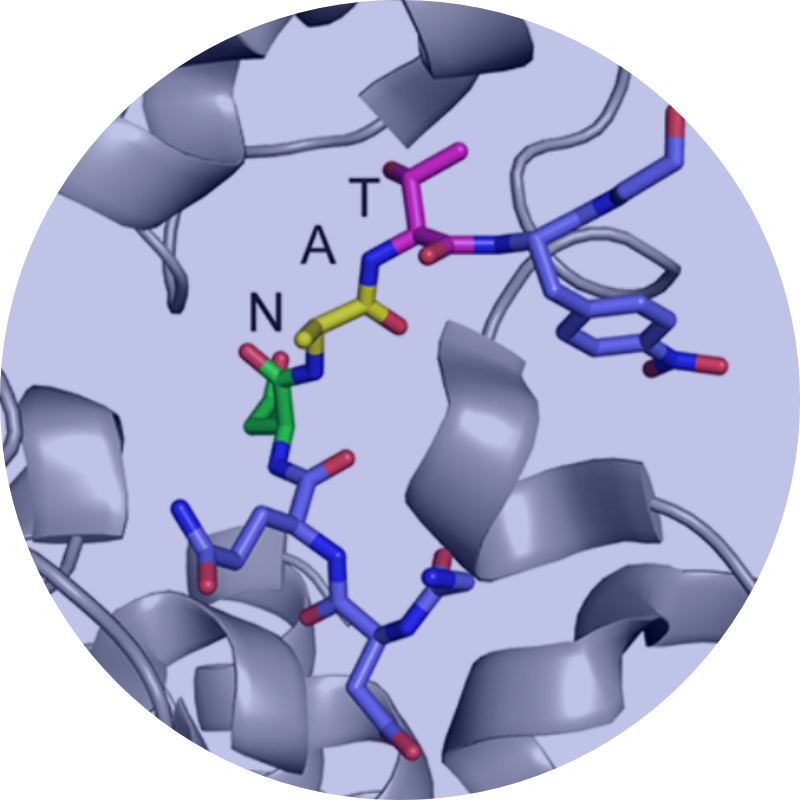
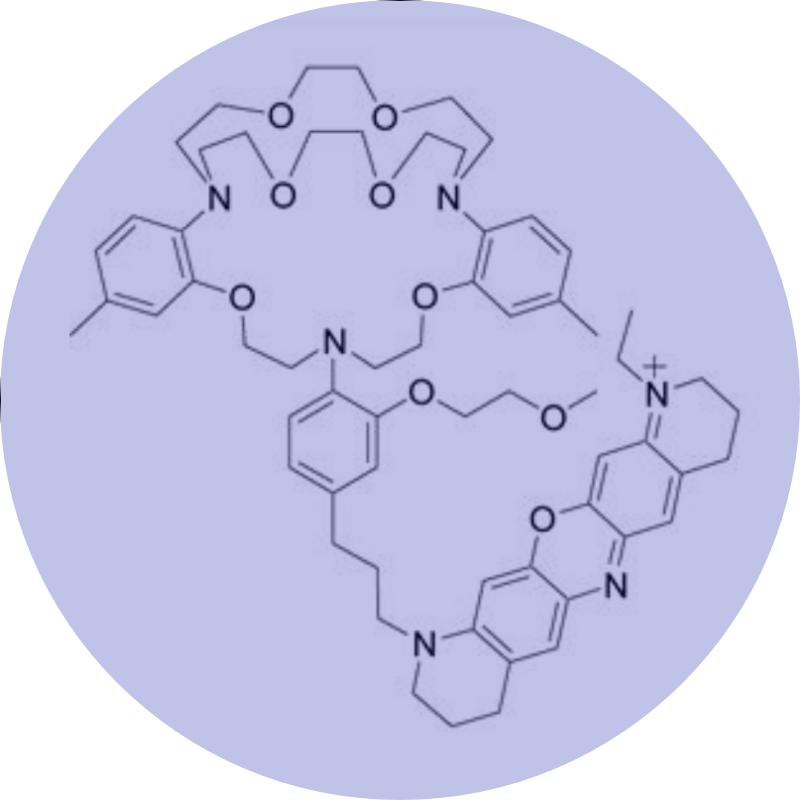
Our long-term research goal is to elucidate the molecular underpinnings of ion channel complex proteins in both healthy and diseased tissues.
x
Research Areas
Want to learn more?
Check out our YouTube channel!
Commonly-Used Techniques
Electrophysiology, biochemistry (especially for membrane proteins), imaging, synthetic organic chemistry, and glycobiology techniques.