Research
Our lab is interested in cell division events, ranging from cell cycle arrest, mitosis, and cytokinesis. Our primary interest is the centrosome and the midbody in their functions during cell life. Both are non-membranous organelles which can rapidly change their protein composition.
The centrosome, also known as microtubule organizing center (MTOC), may actively recruit proteins and mature into spindle pole during mitosis. We recently found out mechanisms how spindle pole protein, pericentrin (PCNT), controls spindle orientation, a process, which might be critical for asymmetric cell division, differentiation, and cyst formation. Mutations in pericentrin are known to cause primary dwarfism, and we explore the phenotype of this rare disorder using pericentrin null mouse model. For example, we observe heart defects, similar to human patients, with mis-orientated mitotic spindles, which eventually lead to defects in septum (model 1). Interestingly, components of recycling endosomes, like Rab11, are important for spindle orientation. Moreover, we now know that some cilia proteins, like IFT88, may operate at the spindle pole and define its orientation. These studies from our lab point to the centrosome as a spindle orientation controlling center.
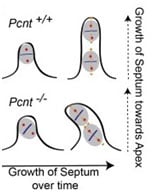
Model 1. Mis-oriented mitotic spindle in PCNT-/- mice contribute to heart septum defects.
The midbody defines the cut of cytokinetic bridge between two daughter cells during the final step in cell division, cytokinesis. Active transport machinery is required to mediate this process. The number of centrosome proteins can be found at the midbody as it is formed, but we wish to know whether there is direct transport of these proteins from the centrosome to the midbody. We are also interested in later destiny of the midbody, particularly, in the phenomenon of their accumulation within the cell. We found out that this accumulation occurs in certain cell types, like stem cells, mainly due to evasion of midbody from autophagy-mediate degradation (Model2). The accumulation of midbody is related to cancer, and the mechanisms of this phenomenon are an active area of our research.
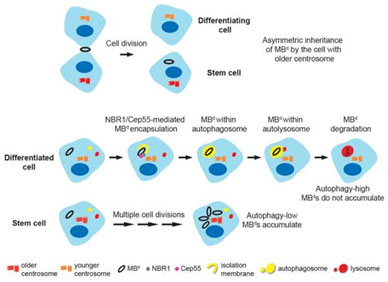
Model 2. Midbody accumulation in stem cells is due to evasion from autophagy.
