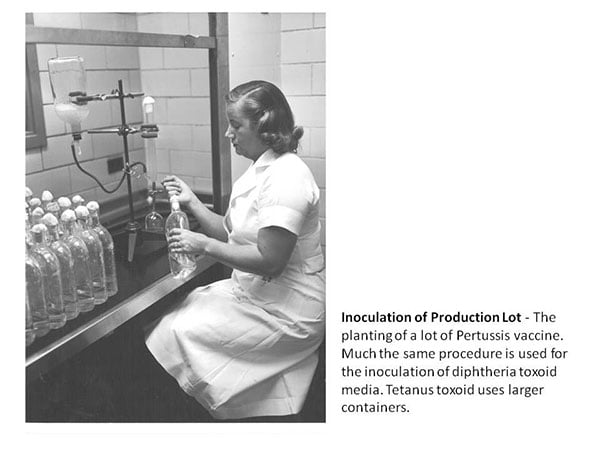
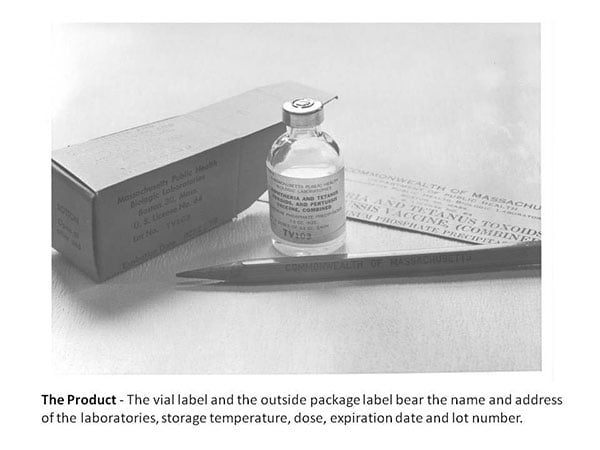
History
The Commonwealth of Massachusetts Board of Health established MassBiologics in 1894 to make diphtheria antitoxin at Harvard's Bussey Institute in Jamaica Plain. The first Director of MassBiologics was the renowned American Scientist, Theobald Smith. In 1914, MassBiologics introduced Diphtheria vaccine into general use in Massachusetts. MassBiologics was granted its first FDA License in 1917 (U.S. License No. 64), making it the 4th oldest biologic manufacturer still in operation in the U.S. today (license number 1779 was granted in 2007 following a name change). Other biologic products developed and manufactured at MassBiologics during the first half of the 20th century were: Smallpox vaccine (1904), typhoid vaccine (1912), the Schick test for diphtheria immunity (1915), anti-pneumococcus serum (1917), scarlet fever antitoxin (1925) and toxin for the Schick test (1936), Tetanus vaccine (1941), Pertussis vaccine (1949) and, in 1950, the first "combination vaccine", Diphtheria-Tetanus-Pertussis vaccine.
In 1941, MassBiologics began distributing Albumin (human) and, in 1942, Immune Globulin manufactured by the Cohn cold-ethanol process developed by Dr. Edwin Cohn at Harvard. MassBiologics was Dr. Cohn's pilot laboratory for this process, which later became the standard process used throughout the world for the manufacture of plasma derivatives. In the last 50 years, MassBiologics pioneered a number of special immune globulins for the prevention of specific diseases. These include Tetanus Immune Globulin (1962), Rh Immune Globulin for prevention of Rh hemolytic disease of the newborn (1970), Hepatitis B Immune Globulin (1972), Varicella Zoster Immune Globulin (1981), Cytomegalovirus Immune Globulin (1990) and Respiratory Syncytial Virus Immune Globulin (1996).
Click here for more information about the history of MassBiologics: http://massbiologicshistory.umassmed.edu