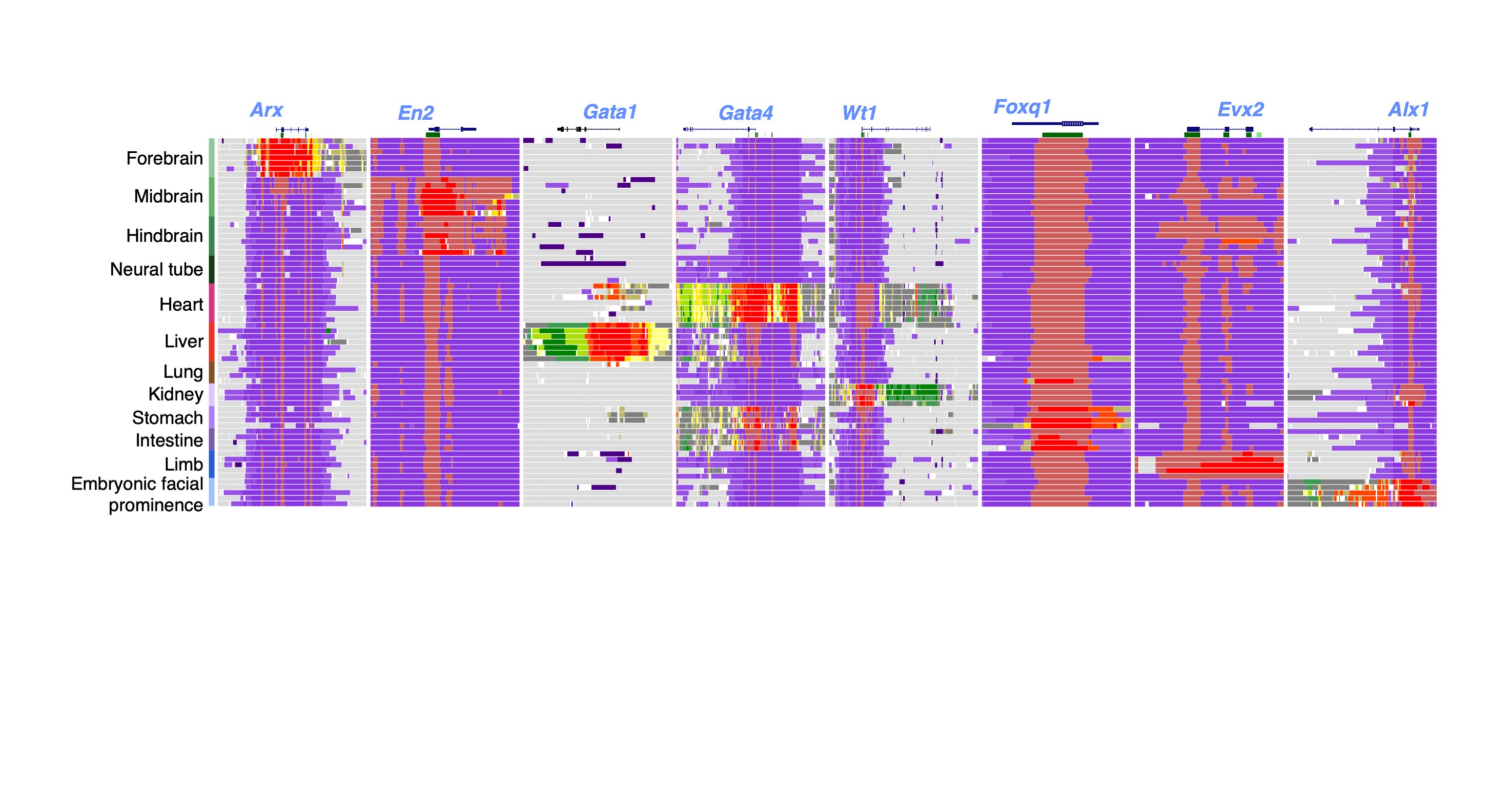
Gene Regulation
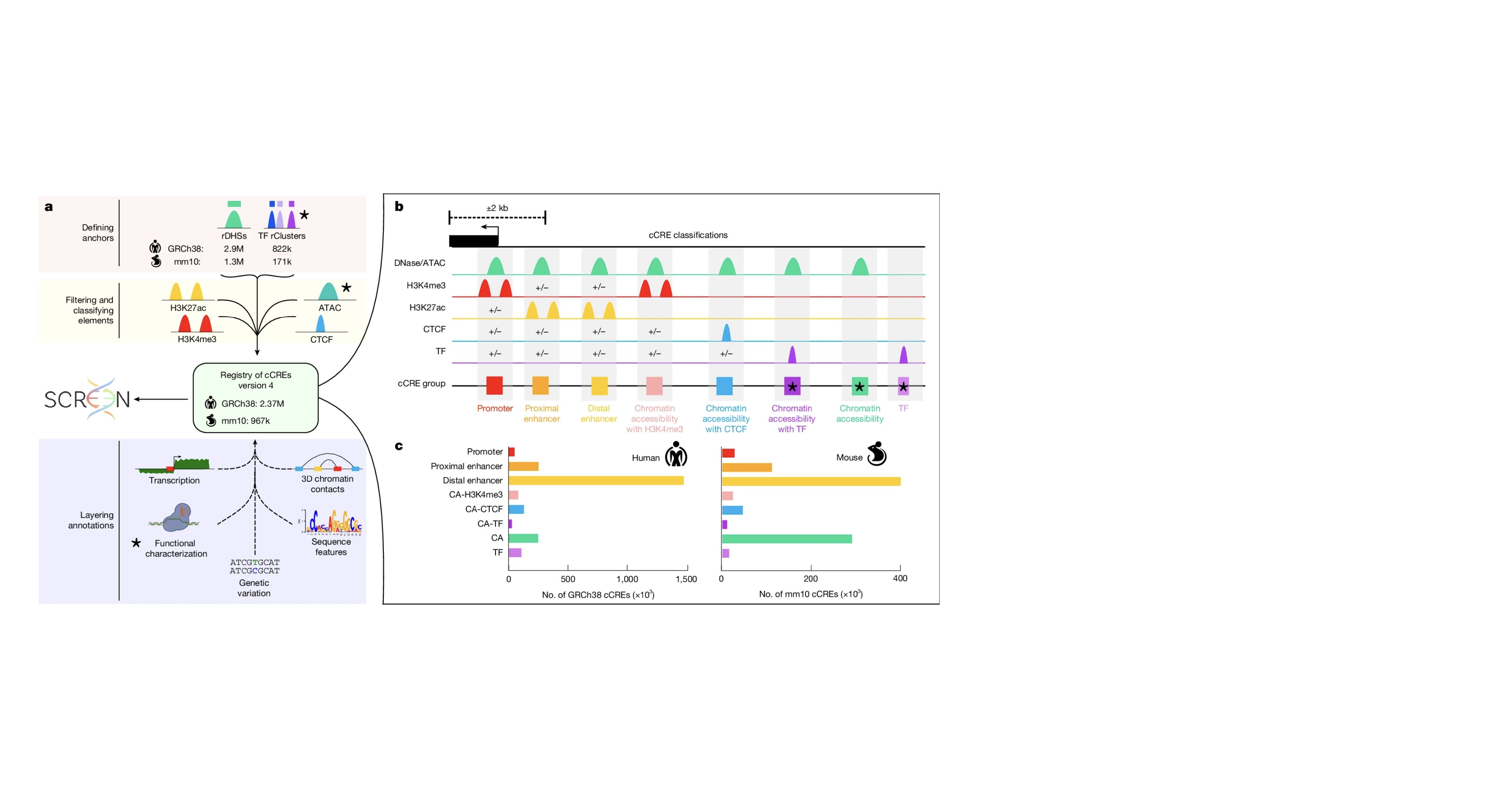
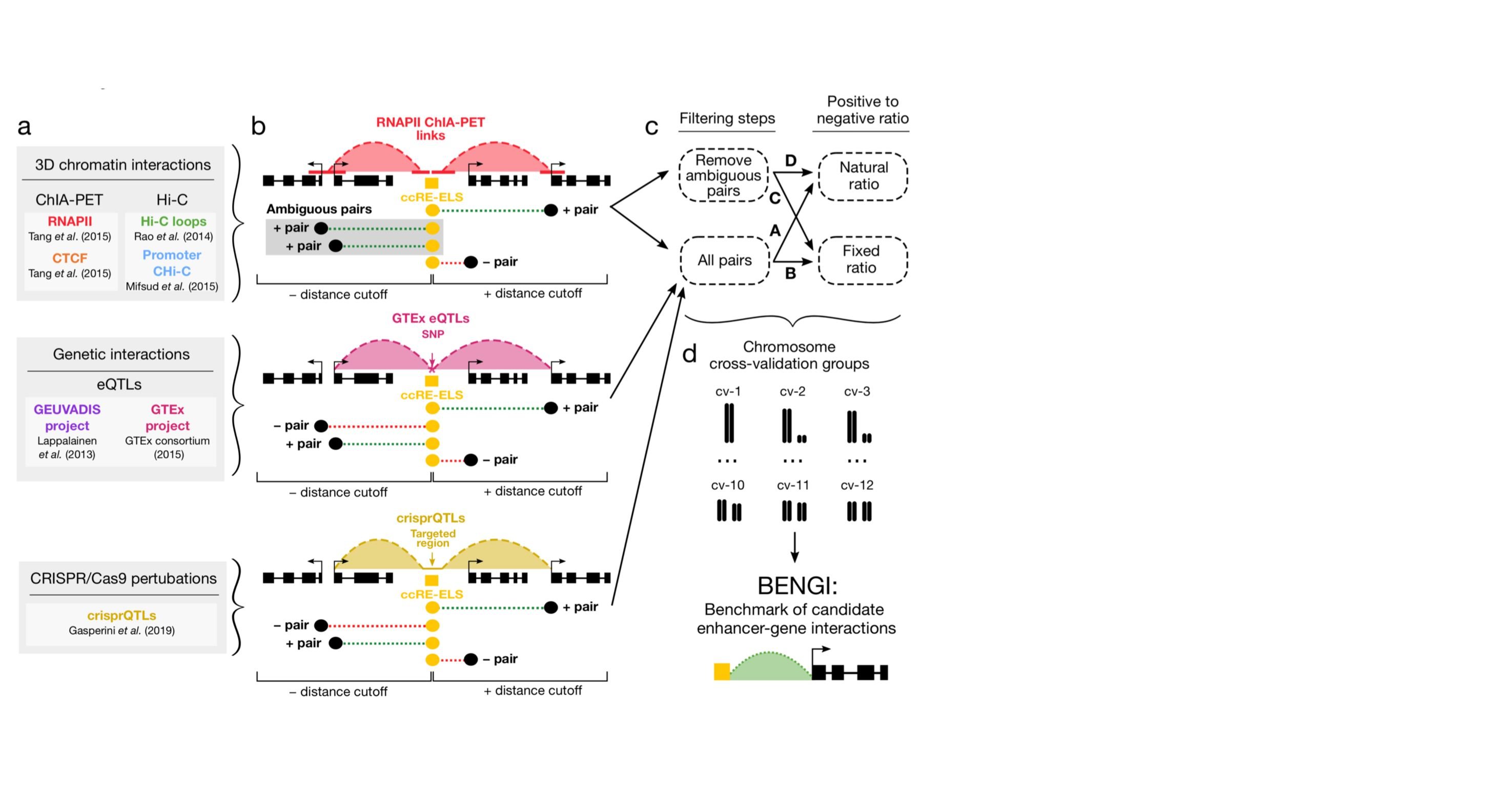
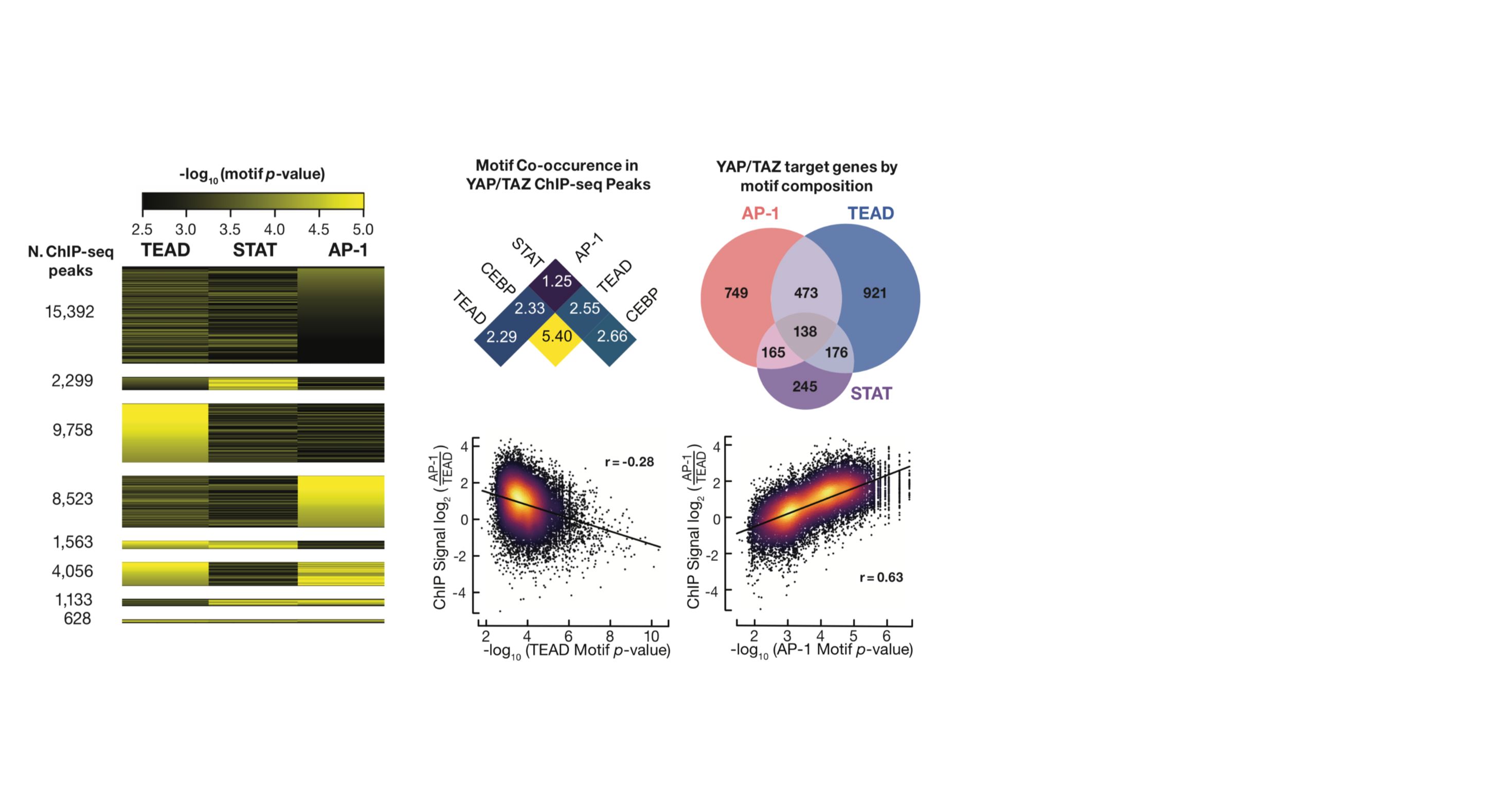
As members of the ENCODE and psychENCODE consortia, we study the molecular mechanism of gene regulation by integrating genomic, epigenomic, and transcriptomic data. Furthermore, we study how genetic variations in the human population affect gene regulation and susceptibility for diseases. We build and apply computational algorithms for analyzing a wide variety of genome-wide deep sequencing data such as ChIP-seq, RNA-seq, DNA-seq, DNase-seq, DNAme-seq, DNA replication, ribosomal profiling, etc.
Protein Docking
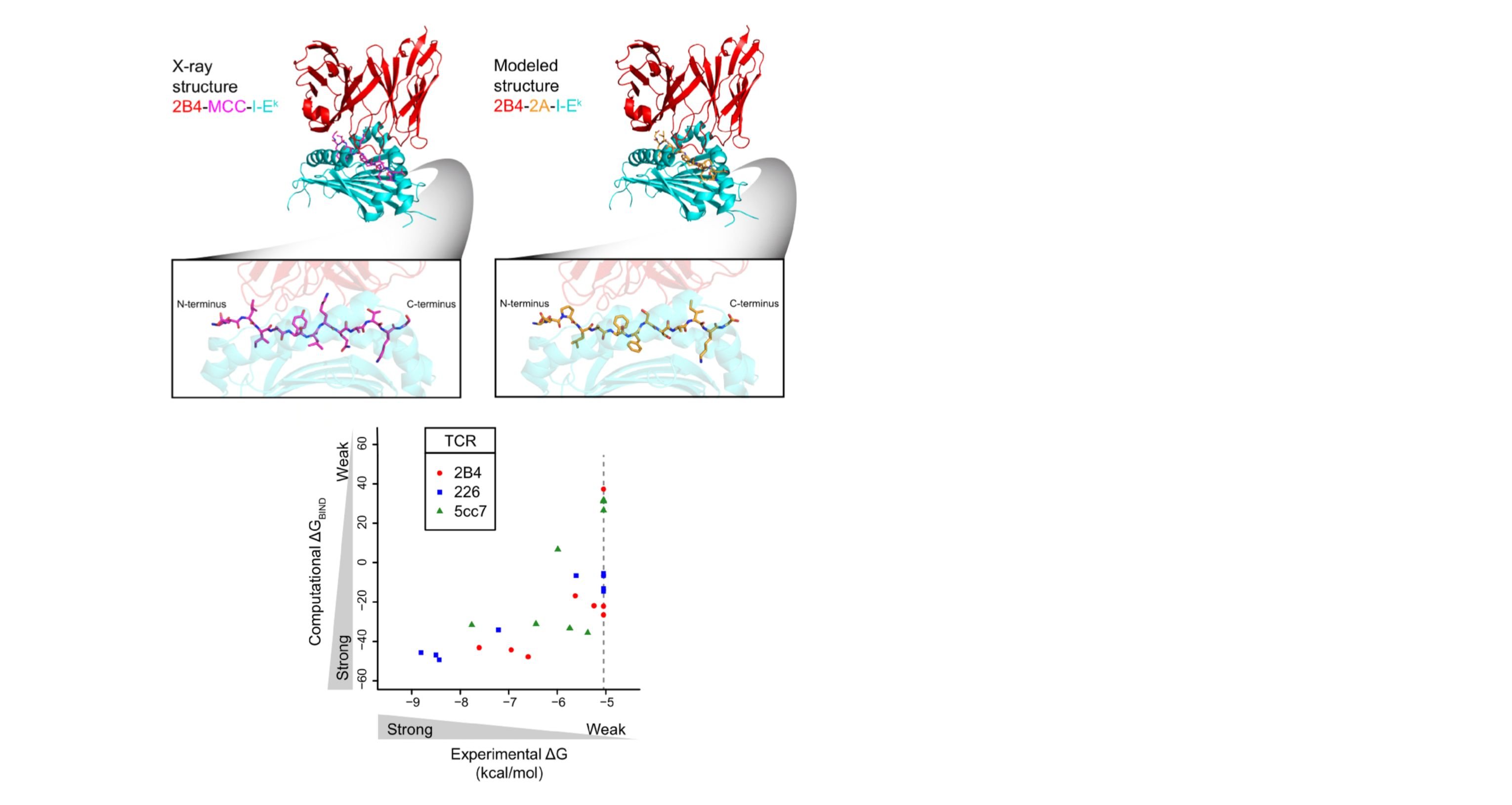
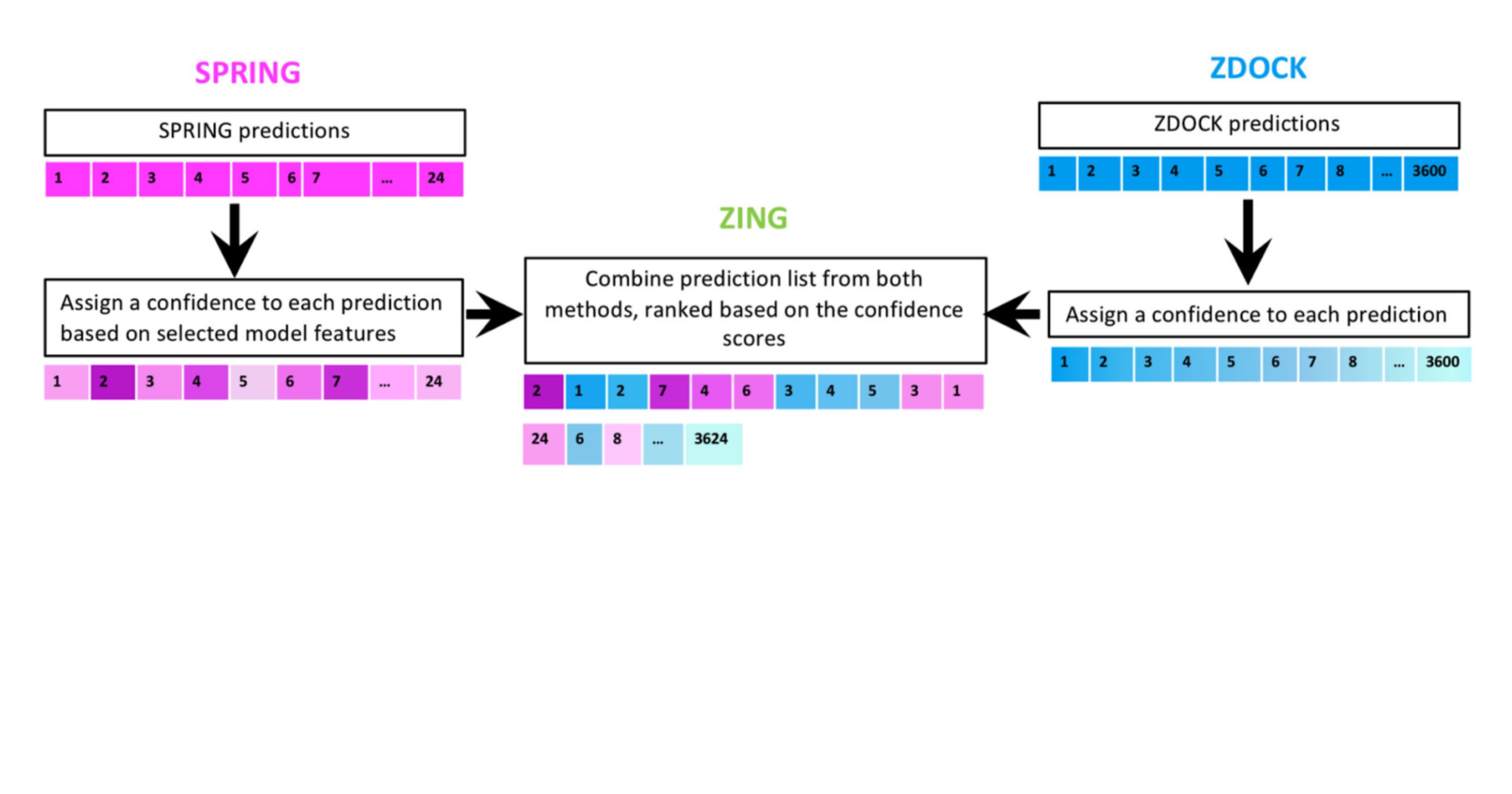
We develop methods to compute binding affinities between protein molecules. Combining this ability with a fast Fourier transform-based search algorithm, we develop computational methods for predicting protein-complex structures. We take a multiple-stage approach, i.e., we have developed an initial-stage algorithm ZDOCK to perform an exhaustive search in the translational and rotational space, and refinement algorithms for structure refinement and reranking. We design T cell receptors with enhanced binding affinity or altered binding specificity.
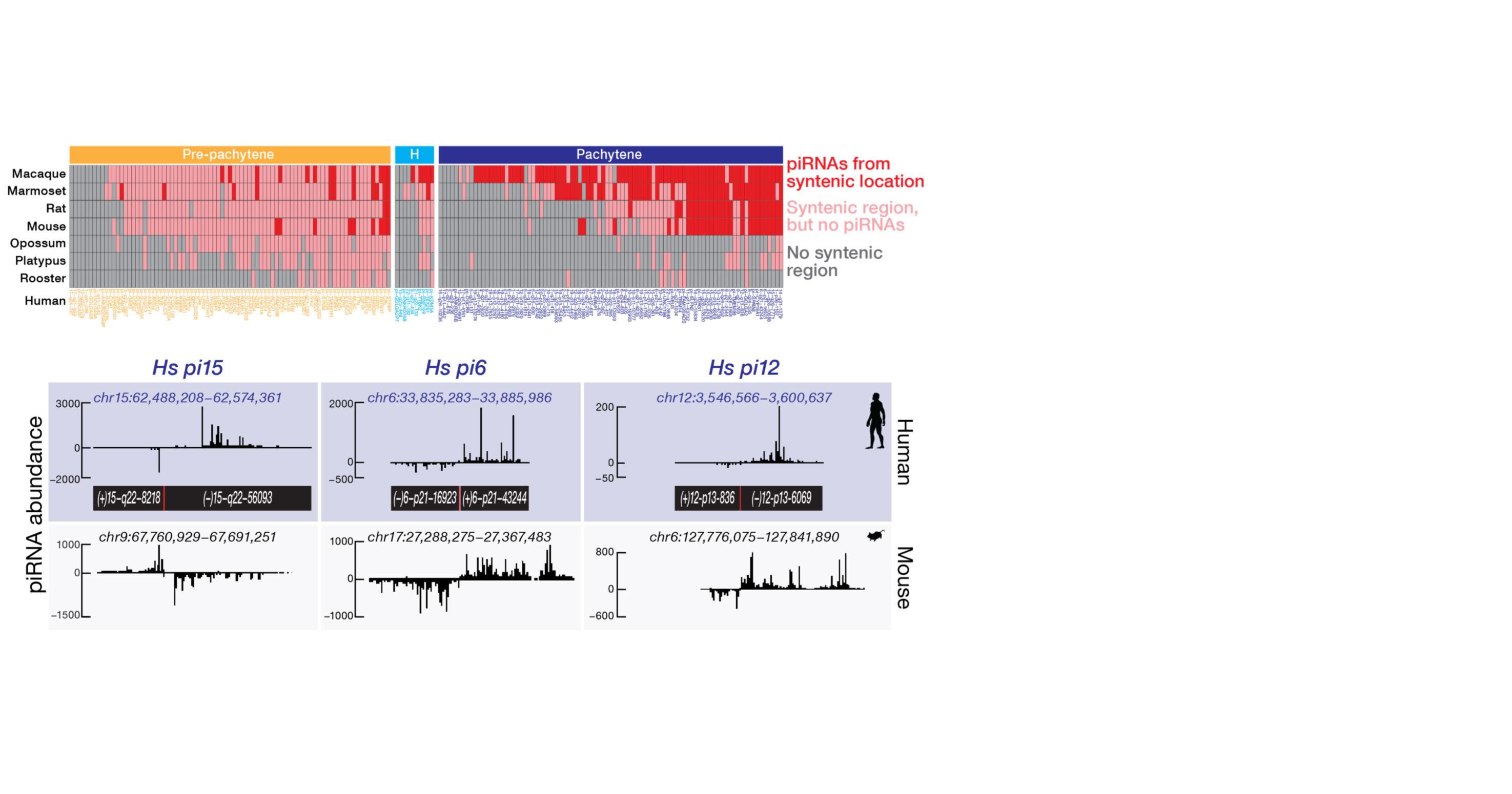
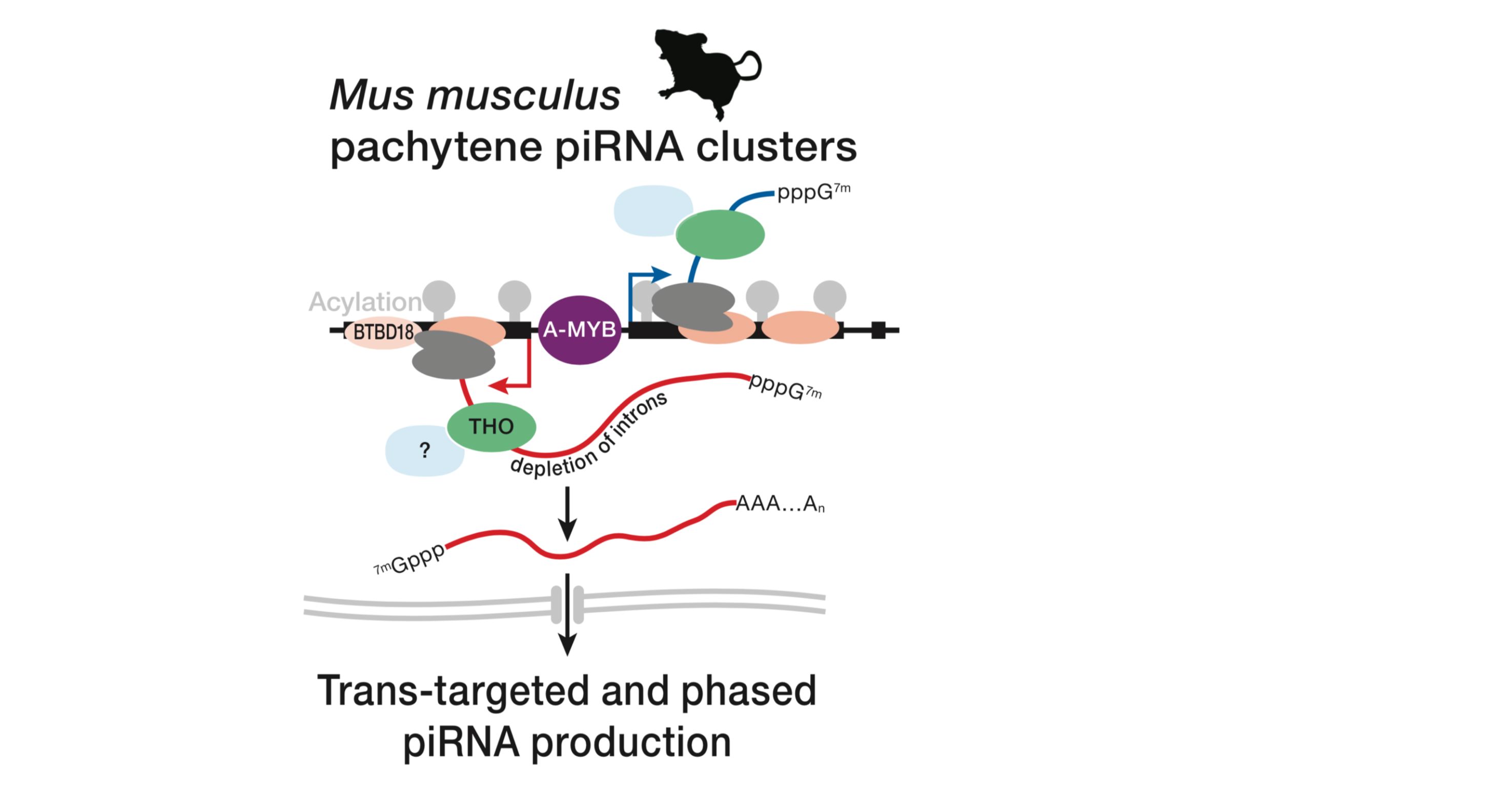
Small Silencing RNAs
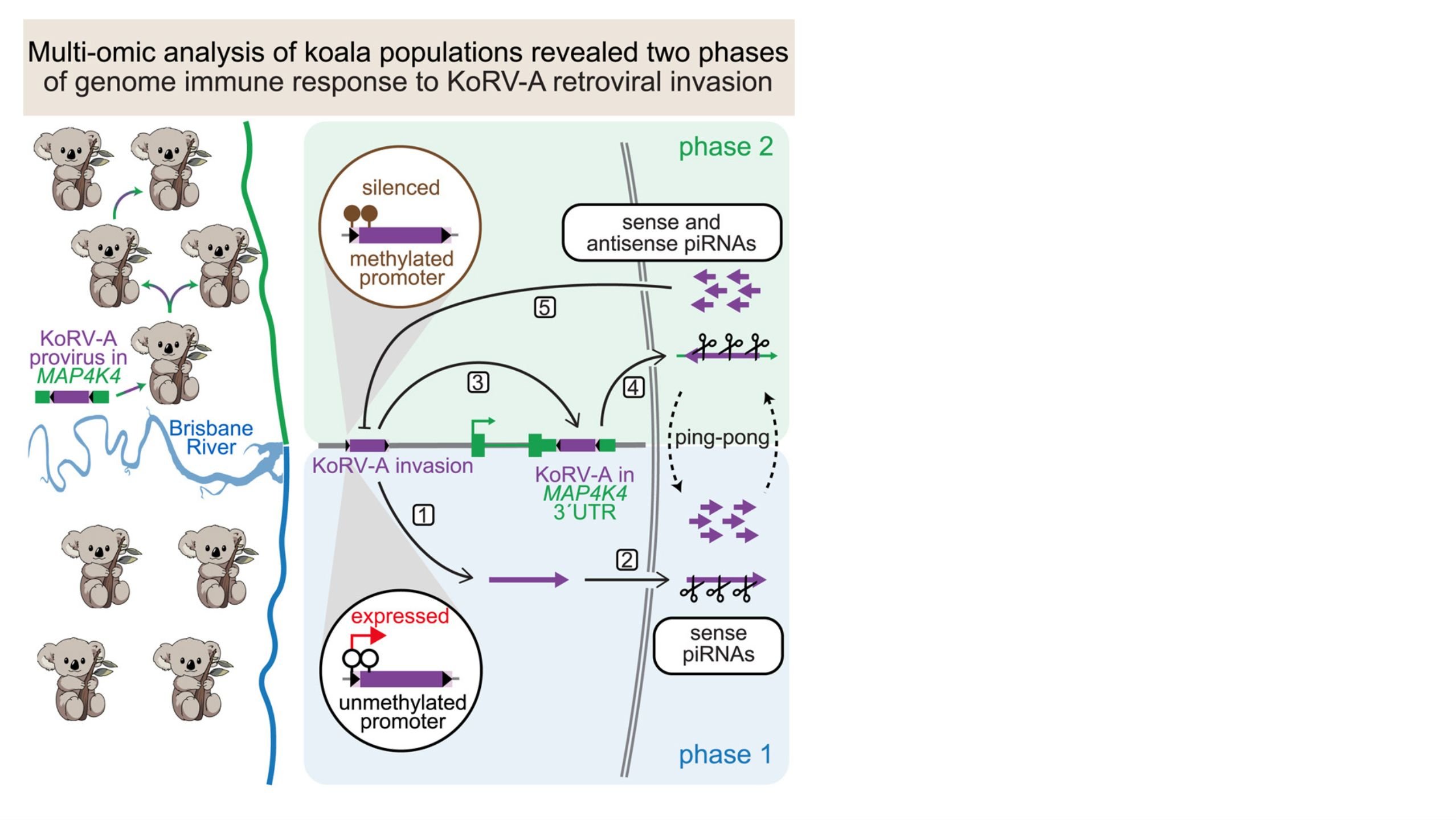
We develop computational methods to understand the biogenesis and regulatory mechanisms of small silencing RNAs (microRNAs or miRNAs, small silencing RNAs or siRNAs, and PIWI-interacting RNAs or piRNAs). We build computational pipelines to analyze diverse high-throughput sequencing data (RNA-seq, DNA-seq, small RNA-seq, ChIP-seq of transcription factors and histone modifications) upon perturbing small silencing RNA pathways.